A Phase 2b/3 Double Blind Safety and Efficacy Study of Injectable Cabotegravir Compared to Daily Oral Tenofovir Disoproxil Fumarate/Emtricitabine (TDF/FTC), for Pre-Exposure Prophylaxis in HIV-Uninfected Cisgender Men and Transgender Women who have Sex with Men

HPTN Primary Paper - NEJM, Aug 11, 2021
NEJM Editorial - Quarraisha Abdool Karim
Laboratory Paper, The Journal of Infectious Diseases, Aug 12, 2021
*** 7 July 2020 ***
HPTN 083 Study Demonstrates Superiority of Cabotegravir for the Prevention of HIV
Press Release Press Coverage July 16 Public Webinar
What is HPTN 083?
HPTN 083 is the first study to compare the efficacy of CAB LA to daily oral TDF/FTC for HIV PrEP. HPTN 083 enrolled 4,570 cisgender men who have sex with men (MSM) and transgender women (TGW) who have sex with men at 43 sites in Argentina, Brazil, Peru, United States, South Africa, Thailand and Vietnam.
 Why is HPTN 083 important?
Why is HPTN 083 important?
For many people, taking a daily pill can be challenging. The development of safe and effective long acting alternative agents for HIV PrEP would increase HIV prevention choices and help those who find taking a daily pill challenging. Some people also may find periodic injections to be a more discreet form of HIV PrEP than daily pills and may prefer CAB LA for that reason.
What happened during the study?
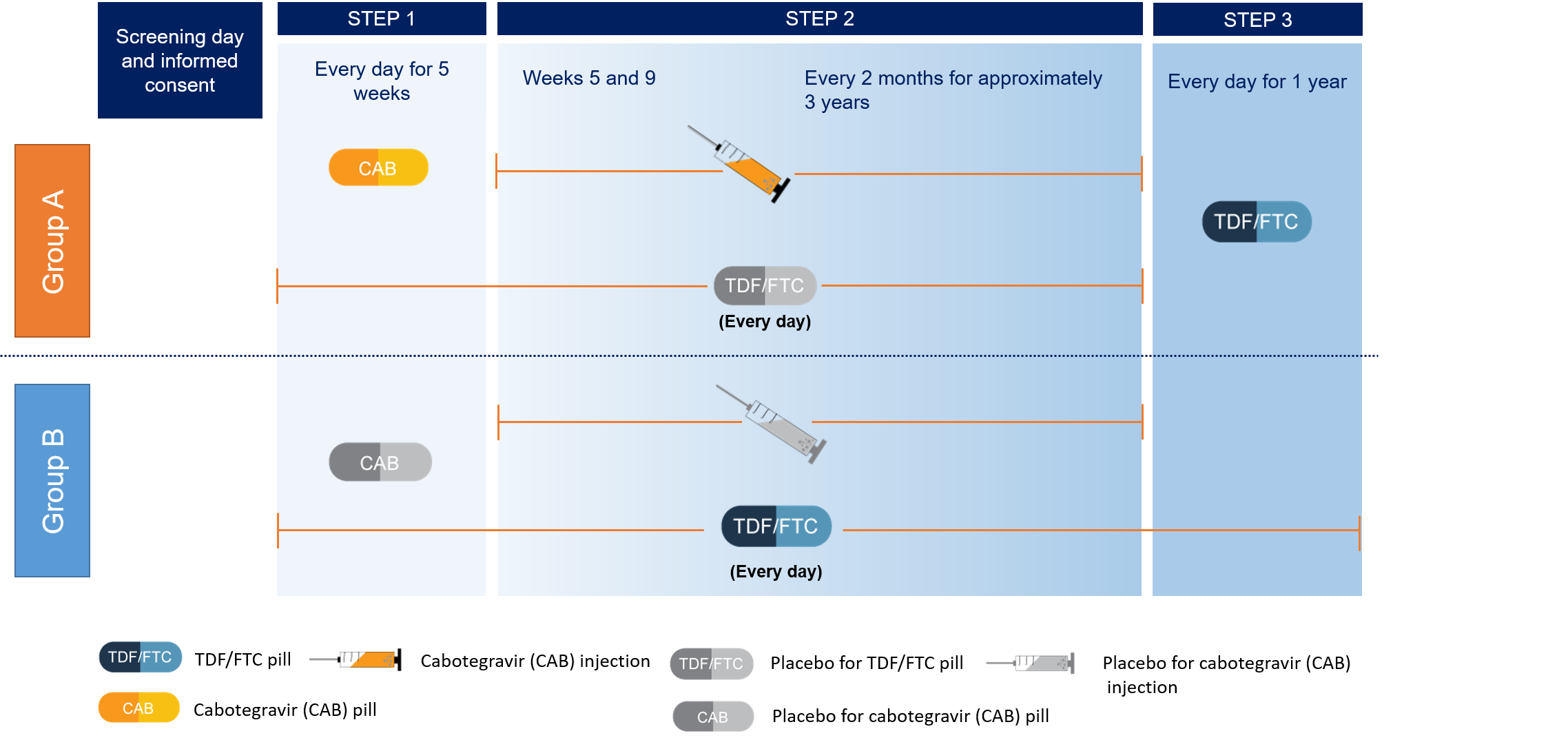
Participants were assigned randomly (by chance) to either the CAB or oral TDF/FTC group. Neither the participants nor the study team knows who was in which group. Participants in each group received both injections and oral tablets – each participant received one active drug and one placebo (no active drug) in order to maintain the blinded nature of the study. Participants were randomized to one of two study arms and included three steps: Step 1 consisted of 5 weeks of daily oral CAB and a TDF/FTC placebo or 5 weeks of daily oral TDF/FTC and an oral CAB placebo; Step 2 consisted of 148 weeks of intramuscular CAB LA 600 mg every 8 weeks plus daily oral TDF/FTC placebo or 148 weeks of daily oral TDF/FTC plus an intramuscular CAB LA placebo every 8 weeks ; and Step 3 consisted of an open-label daily oral TDF/FTC for 48 weeks after participants completed Step 2.
What were the results of the study?
 On May 14, 2020 a Data and Safety Monitoring Board (DSMB) reviewed HPTN 083 study data and recommended that the blinded part of the study be stopped early for successfully meeting its specified objectives. The study results showed that CAB LA, administered every eight weeks, provided high efficacy compared to TDF/FTC. A total of 50 incident HIV infections occurred in HPTN 083, with 38 incident HIV infections in the TDF/FTC arm (incidence rate 1.21%) and 12 incident HIV infections in the CAB arm (incidence rate 0.38%): in other words, approximately three times the number of incident HIV infections were in the TDF/FTC arm than in the CAB arm. The study sponsor, the U.S. National Institute of Allergy and Infectious Diseases (NIAID), approved the decision to stop the blinded part of the study.
On May 14, 2020 a Data and Safety Monitoring Board (DSMB) reviewed HPTN 083 study data and recommended that the blinded part of the study be stopped early for successfully meeting its specified objectives. The study results showed that CAB LA, administered every eight weeks, provided high efficacy compared to TDF/FTC. A total of 50 incident HIV infections occurred in HPTN 083, with 38 incident HIV infections in the TDF/FTC arm (incidence rate 1.21%) and 12 incident HIV infections in the CAB arm (incidence rate 0.38%): in other words, approximately three times the number of incident HIV infections were in the TDF/FTC arm than in the CAB arm. The study sponsor, the U.S. National Institute of Allergy and Infectious Diseases (NIAID), approved the decision to stop the blinded part of the study.
After a more extensive analysis of the interim study data, the regimen containing CAB LA was found to be statistically superior to daily oral TDF/FTC for PrEP among the cisgender men and transgender women who have sex with men enrolled in HPTN 083. A total of 52 incident HIV infections occurred, with 13 incident infections in the CAB arm (incidence rate 0.41%) and 39 incident infections in the TDF/FTC arm (incidence rate 1.22%). The hazard ratio for the CAB versus TDF/FTC arms is 0.34 (95% CI 0.18-0.62), corresponding to a 66% reduction in incident HIV infections in study participants given CAB compared to TDF/FTC. These results were presented at AIDS 2020.
Study Documents
HPTN 083 Version 6.0
HPTN 083 Version 5.0
HPTN 083 Version 4.0
- HPTN 083 Protocol V4 – 10 February 2021
- HPTN 083 Summary of Changes from V3 to V4– 10 February 2021
- HPTN 083 V4.0 CM #1 - 3 March 2021
- HPTN 083 V4.0 LoA #1 - 25 April 2021
- HPTN 083 V4.0 LoA #3 - 1 December 2021
HPTN 083 Version 3.0
- HPTN 083 Protocol V3 – 31 October 2019
- HPTN 083 Summary of Changes from V2 to V3 – 31 October 2019
- HPTN 083 V3.0 LoA # 1 - 19 May 2020
- HPTN 083 V3.0 LoA # 2 - 1 July 2020
- HPTN 083 V3.0 LoA #3 - 23 July 2020
- HPTN 083 V3.0 CM #4 - 5 February 2021
- HPTN 083 V3.0 CM #3 - 4 May 2020
- HPTN 083 V3.0 CM #2 - 2 April 2020
- HPTN 083 V3.0 CM #1 - 10 Feb 2020
HPTN 083 Version 2.0
HPTN 083 Version 1.0
- HPTN 083 Protocol V1.0 - 2 Feb 2016
- HPTN 083 V1.0 LoA #4 - 14 Dec 2017
- HPTN 083 V1.0 LoA #3 - 10 Nov 2016
- HPTN 083 V1.0 LoA #2 - 26 July 2016
- HPTN 083 V1.0 LoA #1 - 24 May 2016
- HPTN 083 V1.0 CM #1 - 13 October 2017
- HPTN 083 V1.0 CM #2 - 19 October 2017
➤ HPTN 083 SSP
(Click to Expand)
Section 1: Introduction
Section 2: Protocol
Section 3: Document Requirements
Section 4: Recruitment, Screening, and Enrollment
Section 5: Study Procedures Overview
Section 6: Visit Checklists
Section 7: Participant Retention
Section 8: Study Product Considerations
Section 9: Clinical Considerations
Section 10: Adverse Event Reporting and Safety Monitoring
Section 11: Laboratory and Specimen Management Procedures
Section 12: Counseling Considerations
Section 13: Data Management
Section 14: Computer Assisted Self-Interview (CASI)
Section 15: Reporting Plan
Section 16: Data Communiqués
Appendix 1: Manual of Operations
Appendix IIA: HPTN 083 Record of Dispensation of Participant-Specific Study Product to Non-Pharmacy Staff
Appendix IIB: HPTN 083 Record of Return of Participant-Specific Study Product by Non-Pharmacy Staff
Appendix III: Schedule of Forms and CASI Surveys
Appendix IV: Participant Transfer and Receipt Process within Medidata Rave
Appendix V: Guidance for the Management of “Discordant/discrepant” HIV Testing Results – HPTN 083 and 084
Appendix VI: Emergency Unblinding by CRS IoR for Medical Reasons
Appendix VII: SSP Manual Updates Per LoA #1 to Version 3.0 of the Protocol
Appendix VIII: SSP Manual Updates Per Version 5.0 of the Protocol
Appendix IX: SSP Manual Updates Per the Open Label Extension (OLE) in Appendix V of Version 6.0 of the Protocol
Press
- December 2016: HIV Prevention Trials Network Launches HPTN 083
- May 2020: Long-acting injectable cabotegravir is highly effective for the prevention of HIV infection in cisgender men and transgender women who have sex with men
Presentations
- Community Educators Training
- Study Specific Training Community Educator Breakout: Retention Strategies
- Study Specific Training Community Educator Breakout: Tracking
- AIDS 2020: HPTN 083 Final Results
Community Presentations for HPTN 083 Results (May 2020)
- English Speakers
- Portuguese Speakers
- Spanish Speakers
- Thai Speakers
- Vietnamese Speakers
More Study Documents
You must request access to private documents (i.e. SSPs) in Microsoft Teams. You can request access to the Team by emailing Jeff Webb (jwebb@fhi360.org).
You will need a Microsoft account to log in to Team. Click here for instructions on setting up your Microsoft account and accessing the Teams platform.
Study Details
To evaluate the safety and efficacy of the injectable agent, cabotegravir (CAB LA), for pre-exposure prophylaxis (PrEP) in HIV-uninfected cisgender men and transgender women who have sex with men (MSM and TGW)
Multi-site, double blind, two-arm, randomized (1:1), controlled non-inferiority trial of the efficacy of CAB LA compared to daily oral tenofovir disoproxyl fumarate (TDF)/emtricitabine (FTC) for HIV prevention
HIV-uninfected MSM and TGW at risk for acquiring HIV infection, ages 18 or older.
Approximately 4500, 2250 per arm
Approximately 4.5 years total, with individual participants being followed between 1.5 years (for the latest enrolling participants) to 4.5 years (for the earliest enrolling participants). Accrual will require approximately 130 weeks. In Step 1, participants will receive oral tablets for 5 weeks. In Step 2, participants will receive injections (as a single injection at two time points 4 weeks apart and every 8 weeks thereafter) and daily oral tablets. Step 2 will be continued until the required number endpoints is reached, estimated to be approximately when the final participant reaches 60 weeks on Step 2 (week 65 for the final participant). Participants will be all simultaneously unblinded at the conclusion of Step 2. In Step 3, all participants will receive open-label daily oral TDF/FTC for up to 48 weeks. Participants will therefore be followed between 113 weeks to 233 weeks (between 65 and 185 weeks on blinded study medication and up to 48 weeks on open-label daily oral TDF/FTC). All participants will be transitioned to locally-available HIV prevention services, including services for PrEP, if available, at the end of their participation in the study.
Once randomized to one of two arms, participants will move through the following steps:
Step 1:
Arm A – Daily oral CAB (30 mg tablets) and oral TDF/FTC placebo for five weeks
Arm B – Daily oral TDF/FTC (300 mg/200 mg fixed-dose combination tablets) and oral CAB placebo for five weeks
A participant that becomes HIV-infected during Step 1 of the study will permanently discontinue study product and will be terminated from the study, and referred for HIV-related care.
Step 2:
Arm A – CAB LA (600 mg as a single intramuscular [IM] injection at two time points 4 weeks apart and every 8 weeks thereafter) and daily oral TDF/FTC placebo.
Arm B – Daily oral TDF/FTC (300/200 mg fixed-dose combination tablets) and IM placebo at two time points 4 weeks apart and every 8 weeks thereafter (matching vehicle, identical volume as active injectable product in Arm A).
This step will continue until the required number of endpoints is reached.
A participant that becomes HIV-infected during Step 2 of the study will permanently discontinue study product, be placed on immediate suppressive ART, and will be followed at quarterly intervals for 52 weeks after their last injection prior to diagnosis of HIV in order to test for safety parameters, as well as CD4 cell count and HIV viral load. After 52 weeks, they will be terminated from the study and transitioned to continued HIV-related care.
Step 3:
Both arms: Open-label daily oral TDF/FTC no later than 8 weeks after the last injection (in order to cover the pharmacokinetic (PK) tail for Arm A participants), for up to 48 weeks. Participants will then transition to locally-available HIV prevention services, including services for PrEP, if available.
A participant with confirmed HIV infection during Step 3 will be followed at least for the duration of Step 3, with possible additional assessments and follow-up to be determined by the Clinical Management Committee (CMC).
• To compare HIV incidence among participants randomized to oral CAB/CAB LA (oral lead in and injections) vs. oral TDF/FTC (Steps 1 and 2)
• To compare the safety of oral CAB/CAB LA vs. oral TDF/FTC
• To compare HIV incidence among participants receiving CAB LA injections vs. oral TDF/FTC (each step independently and all steps in aggregate)
• To compare HIV incidence among participants receiving CAB LA injections vs. oral TDF/FTC (Steps 1, 2, and 3 combined)
• To compare HIV incidence among participants randomized to CAB LA injections vs. oral TDF/FTC while taking open label TDF/FTC (Step 3 only, descriptive)
• To compare the change in risk of HIV acquisition between CAB and oral TDF/FTC strategies (Arm A and Arm B) as participants progress from Step 2 to Step 3
• To compare HIV incidence among the subgroups of participants receiving oral CAB/CAB LA vs. oral TDF/FTC by region, age, race, ethnicity, baseline risk, and gender identity
• To compare changes in renal function, liver function, and bone mineral density (BMD) among participants receiving oral CAB/CAB LA vs. oral TDF/FTC
• To evaluate and compare rates of HIV drug resistance among participants who acquire HIV infection during the study among participants receiving oral CAB/CAB LA vs oral TDF/FTC
• To compare the acceptability of and preferences for CAB LA vs. oral TDF/FTC
• To examine the association between levels of adherence and HIV incidence
• To compare and describe the rates, patterns, and correlates of adherence to CAB LA vs oral TDF/FTC, in aggregate and by psychosocial/demographic variables
• To estimate changes in sexual-risk behavior as measured by self-report and rates of incident gonorrhea, chlamydia, and syphilis in the study population
• To compare the resource utilization and programmatic costs of long-acting injectable PrEP vs. daily oral PrEP vs. no PrEP for HIV uninfected MSM and TGW in the US, Brazil, South Africa and India
• To use mathematical simulation to project the short- and long-term clinical impact, cost projections, budgetary impact, and incremental cost-effectiveness of long-acting injectable PrEP vs. daily oral PrEP vs. no PrEP for HIV uninfected MSM and TGW in the US, Brazil, South Africa and India

